 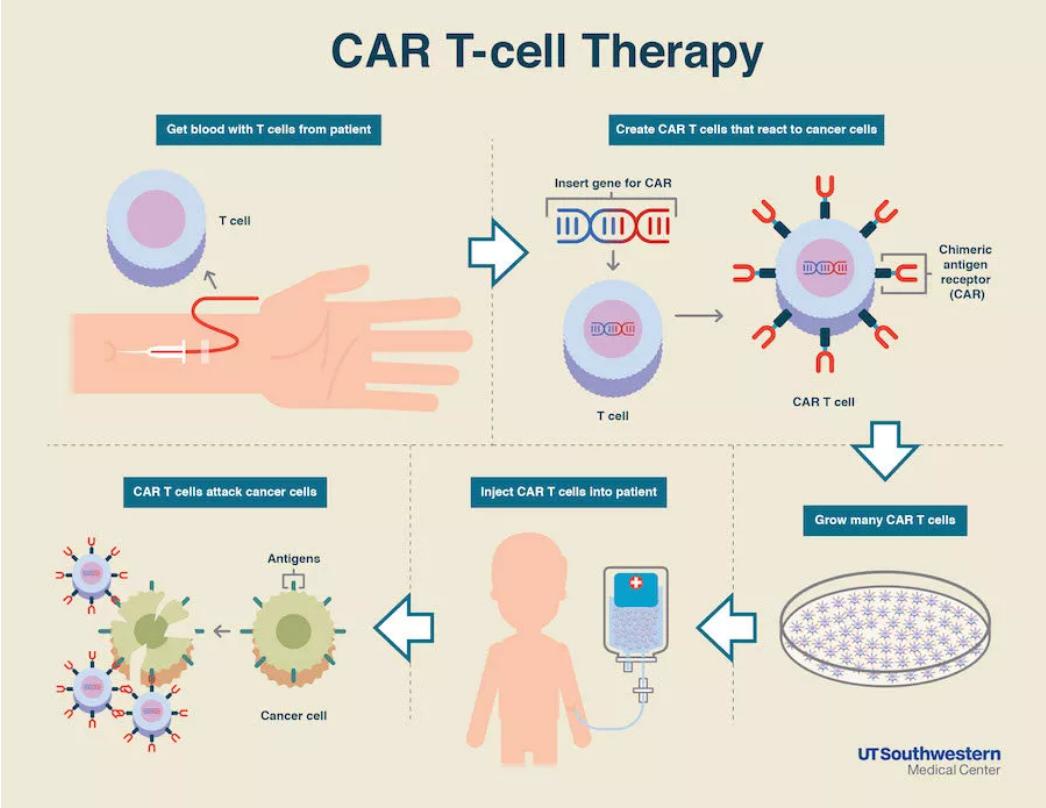
Although CAR-T cells with high specificity are desirable, expression of target antigen in normal cells can lead to unwanted toxicities. Therefore, future studies are warranted to better understand mechanisms for resistance and relapses during CAR-T cell therapy and develop novel strategies to overcome these challenges.ĬARs are synthetic receptors that are developed to interact with target cells with high specificity. Thus, numerous mechanisms may contribute to resistance and relapse during CAR-T cell therapy. Other rare events such as transduction of a tumor cell during manufacturing and subsequent CAR expression in the malignant cell resulting in resistance to CAR-T cell therapy has been also observed ( 24). Experience with CD19 CAR-T cells suggest a significant number of patients can relapse due to loss of CD19 expression ( 11). Antigen-null or antigen-dim cancer cells can arise via various mechanisms, which can contribute to resistance to CAR-T therapy ( 20– 23). 
Loss or decrease in target antigen expression can also affect efficacy of CAR-T cell therapy. Other strategies to mitigate CAR-T cell exhaustion are also in development ( 17– 19). anti-PD-1 or anti-PD-L1) to block PD-1 and PD-L1 interaction between CAR-T cells and cancer cells are being used in clinical trials to restore CAR-T cell function ( 15, 16). Strategies such as use of checkpoint inhibitors (e.g. One of the mechanisms that can lead to loss of function is CAR-T cell exhaustion ( 3, 12). Following infusion, CAR-T cells may fail to proliferate or persist leading to loss of therapeutic response ( 15). It is anticipated that the field of CAR-T cell therapy will continuously grow, and new CAR-T cells will be developed to treat previously incurable human diseases.īased on the data obtained from clinical studies with CD19 CAR-T cell therapies, CAR-T cells can be curative in those patients who respond to the therapy however, not all patients respond in a similar manner and challenges such as resistance and relapses have been observed ( 3, 11– 14). Many other CAR-T cells targeting various antigens other than CD19 are either in pre-clinical development or currently in clinical trials to treat various human diseases such as cancer, infectious diseases, autoimmune diseases, cardiac diseases etc. FDA approval of one BCMA-targeting and several CD19-targeting CAR-T cell therapies for certain relapsed/refractory hematologic malignancies have energized the field ( 2– 8). Although CAR-T cell therapy has been in development for more than two decades ( 1), recent U.S. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
